New multi‑dose pen boosts Zepbound’s edge as Novo Nordisk’s CagriSema trial data disappoints
One pen, four weekly obesity doses: Eli Lilly is using that formula to cement Zepbound’s lead as Novo Nordisk’s next big hope stumbles in trials.
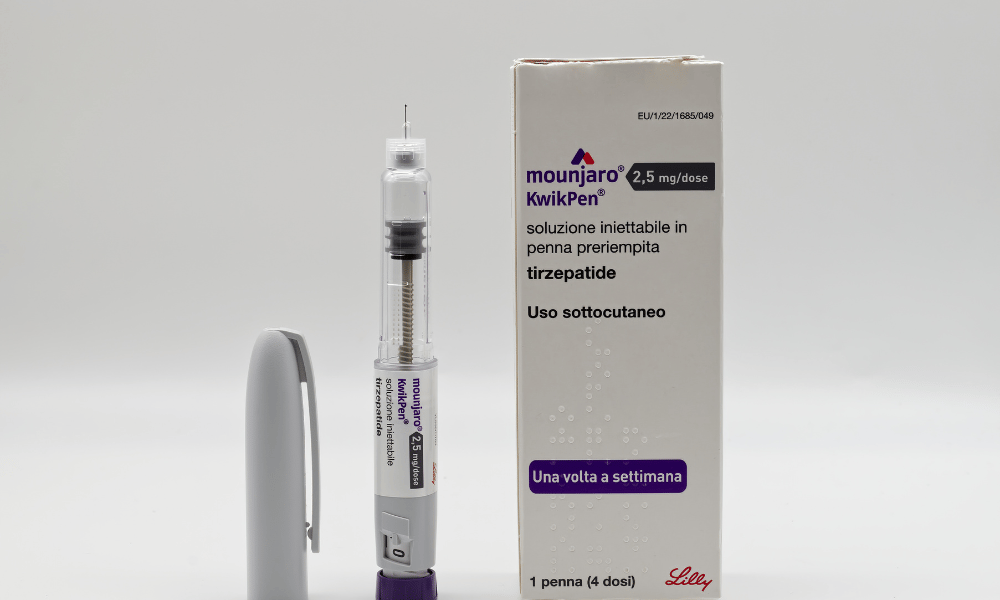
Eli Lilly has received US Food and Drug Administration approval for a four‑dose Zepbound “KwikPen” that delivers a full month of treatment in a single device, according to Reuters.
CNBC reported that the agency approved a label expansion for Zepbound to add the multi‑dose device. Cash‑paying patients can get the KwikPen through Lilly’s direct‑to‑consumer website, LillyDirect, with prices starting at US$299 per month for the lowest dose.
According to Reuters, the multi‑dose injection pen will be available in all six Zepbound strengths – 2.5 mg, 5 mg, 7.5 mg, 10 mg, 12.5 mg and 15 mg – and delivers a full month of therapy in one device.
CNBC reported that patients can use a single KwikPen to take four weekly doses of Zepbound, instead of using a different single‑dose autoinjector each week or drawing the drug from vials into a syringe.
Reuters said Zepbound, approved by the FDA in 2023, is also available as a single‑dose autoinjector and in vials in the United States.
The KwikPen platform is already established.
Eli Lilly uses the same device for its diabetes drug Mounjaro, and the device is available in international regions including the United Kingdom, Australia, the Middle East and Canada.
Demand for Zepbound has surged since it entered the US market in late 2023.
CNBC reported that the drug has “exploded in demand” and helped Eli Lilly capture a majority share of the weight‑loss drug market from Novo Nordisk.
In the company’s fourth quarter, Zepbound generated US$4.2bn in US revenue, a 122 percent increase from the prior year.
Reuters said Zepbound has overtaken Novo Nordisk’s Wegovy as the US weight‑loss market leader based on prescription data.
CNBC reported that LillyDirect has been key to Zepbound’s growth and that launching the KwikPen on the platform could attract more patients.
Novo Nordisk’s latest trial results have reinforced that shift.
Reuters reported that late‑stage data for Novo’s next‑generation obesity drug CagriSema showed a 23 percent reduction in body weight over 84 weeks, compared with 25.5 percent for Eli Lilly’s tirzepatide, the active ingredient in Zepbound and Mounjaro.
The trial was designed to show CagriSema could be at least as effective as tirzepatide, but the outcome undercut that goal and appeared to show greater weight loss with Zepbound than in some of Lilly’s own earlier trials, which ran for shorter periods.
The market reaction was immediate.
Novo Nordisk’s shares fell 16 percent while Eli Lilly’s rose 5 percent after the data, as investors questioned Novo’s obesity pipeline and its ability to regain ground.
BMO Capital analyst Evan Seigerman told Reuters, “They literally ran a trial that said that Lilly’s product is better,” adding that Novo has had “twin misses with CagriSema in just over a year.”
Analysts quoted by Reuters said the results strengthen Lilly’s competitive position.
Deutsche Bank wrote that the diabetes and obesity market is “likely to coalesce around Lilly’s portfolio,” while JP Morgan said it would be “difficult for Novo to dislodge market share from Lilly, with Zepbound well entrenched.”
HSBC analyst Rajesh Kumar said the trial “seems to further raise the question of the value proposition of CagriSema in the obesity market landscape.”
Barclays said the outcome likely means Novo will face an uphill battle convincing patients and physicians to choose CagriSema over “more effective, better tolerated Zepbound,” leaving Novo “little to compete on apart from price.”
Novo’s leadership has tried to counter that narrative.
During an investor call, Deutsche Bank analyst Emmanuel Papadakis asked whether CagriSema was now “obsolete” as a competitive upgrade to Wegovy, Reuters reported.
CEO Mike Doustdar responded that, once approved, CagriSema would have the best label data and described Zepbound’s performance as an “abnormality,” while Chief Scientific Officer Martin Holst Lange said patience was needed to see CagriSema’s full potential.
One Novo shareholder quoted by Reuters dismissed management’s explanation as a “lame excuse.”



